Introduction – Discovery – How is it Made?
Hydrogen Peroxide and History – Hydrogen Peroxide and the Future
Hydrogen Peroxide was first dicovered by Louis- Jacques Thenard in 1818 by burning barium salts to create barium peroxides. When the Barium peroxides were put into water to dissolve, Thenard found that hydrogen peroxide was produced.
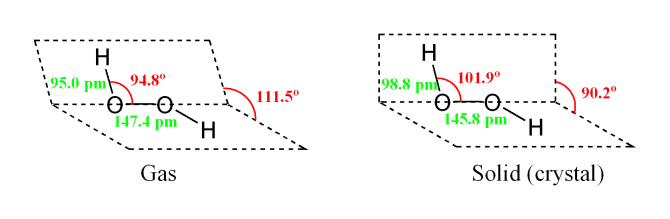
Hydrogen Peroxide is a is a strong oxidizing agent and a weak acid in water solution. The formula is similar to that of water, with an extra atom of oxygen attached, H2O2. It is completely soluble in water. Pure anhydrous hydrogen peroxide is a colorless to pale blue syrupy liquid which decomposes violently into water and oxygen if heated above 80 C. it also decomposes in light and in the presence of metal ions or oxidizable organic materials. One volume of hydrogen peroxide releases ten volumes of oxygen when it decomposes. The most valuable property of hydrogen peroxide is that it breaks down into water and oxygen and therefore does not form any persistent, toxic residual compounds.